2. Xi′an Modern Chemistry Research Institute, Xi′an 710065, China
2. 西安近代化学研究所, 陕西 西安 710065
As one of the most simple furazan ring compounds, 3, 4-diaminofurazan(DAF) features good thermal stability and big density, which make it efficient structural units in the development of high-energy-density materials(HEDMs)[1-5]. DAF has been an important precursor to a series of furazan-based energetic materials[6-10], 3, 4-dinitrofurazan (DNF), 4, 4-dimino-3, 3-azofurazan(DAAzF) are two relatively simple derivatives of DAF, and their relationship are shown in Scheme 1. DNF was firstly reported at 1994[11], having a crystal density of 1.62 g·ml-1, a melting point of 15.8 ℃ and a boiling point of 168.8 ℃. DNF is a high energy explosive and has been exploited the high reactivity of the nitro-groups to nucleophile in the synthesis of a large number of energetic derivatives. DAAzF is another special rich-nitrogen insensitive energetic material for its big positive enthalpy of formation[12]. Many studies showed that the three furazan compounds have excellent properties and good application prospect[13-14].
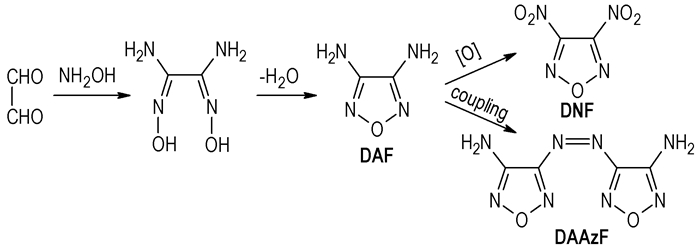
|
Scheme1 Relationship of DAF, DNF and DAAzF |
Energy of combustion and specific heat capacity are twoimportant thermodynamic data and characteristic quantities closely related to energy and structure of material. We researched energies of combustion and specific heat capacities of the three furazan ring compounds to enrich thermochemical database and provide theoretical basis for further application. Meanwhile, the change rule of structure-property for the three compounds was also discussed.
2 Experimental 2.1 SamplesDAF, DNF and DAAzF were synthesized by our research group in Xi′an Modern Chemistry Research Institute. Their purities are more than 99.5%(HPLC). DAF and DAAzF are solid, and DNF is liquid.
2.2 Energy of CombustionEnergy of combustion was determined with an IKA C5000 oxygen-bombcalorimeter (German) in adiabatic pattern. The calorimeter was calibrated with the standard substance benzoic acid having a purity of 99.99%, and each sample was tested with 6 times, The mean value (-26504±147) J ·g-1[15] is very close to the standard value as (-26434±3) J ·g-1 (T=298.15 K)[16], indicating that the measuring system is accurate and reliable. The uncertainty can be obtained by equation Uc=ku, where u is the standard uncertainty (the standard deviation of mean) the coverage factor k is 2, and level of confidence is 0.95.
2.3 Specific Heat CapacitySpecific heat capacity was measured using a Micro-DSCⅢ apparatus (SETARAM, France), with the operating temperature range of 283-333 K, temperature accuracy of 10-4 K, heat flow accuracy of 10-4 mW, and heating rate of 0.15 K ·min-1. The sample mass used for calorimetric measurement was about 200 mg. The reliability of enthalpy measurement was ensured by determinations of the enthalpy of dissolution of KCl (cr) in deionized water at 298.15 K. The result was (17.27±0.07) kJ·mol-1, which was very close to the literature value as (17.24±0.02) kJ·mol-1[17]. The equation of specific heat capacity for standard calcined α-Al2O3 obtained was cp (J·g-1·K-1)=0.184+1.997×10-3 T (283 K < T < 353 K), and the standard molar heat capacity is 79.44 J·mol-1·K-1(298.15 K), which is consistent with the literature value of 79.02 J·mol-1·K-1[18]. The results indicate that calorimetric equipment is reliable and accurate.
3 Results and Discussion 3.1 Energy of CombustionEach sample was tested with 6 times, and the results are listed in Table 1. The constant-volume energies of combustion for DAF, DNF and DAAzF are (-13043±119), (-6863±37), (-12661±54) J·g-1, respectively. The energy of combustion tends to rise with the decrease of oxygen content in molecule, DAF(15.99%)>DAAzF(16.31%)>DNF(49.98%). DAF and DAAzF exhibit greater energy of combustion than DNF, indicating that amino group is an excellent burning group and markedly increase energy of combustion, but nitro group has the opposite influence. Due to the same number of amino group and the approximate nitrogen content (oxygen content), the difference of energy of combustion for DAF and DAAzF is small.
| Tab.1 Determination results for the energies of combustion of the samples |
The standard molar enthalpy of combustion (ΔcHmΘ) was referred to the energy of combustion change of the following idealized reaction equation (1) at T=298.15 K and pΘ=101.325 kPa[19].
| $ {\rm{M(s) + }}\mathit{a}\;{{\rm{O}}_{\rm{2}}}({\rm{g}}) = \mathit{b}\;{\rm{C}}{{\rm{O}}_2}({\rm{g}}) + \mathit{c}\;{{\rm{H}}_2}{\rm{O({\rm{l}}) + }}\mathit{d}\;{{\rm{N}}_2}({\rm{g}}) $ | (1) |
M=DAF:C2H4ON4, a=5/2, b=2, c=2, d=2;
DAAzF:C4H4O2N8, a=4, b=4, c=2, d=4;
Herein, it is necessary to illustrate that DNF is an oxygen-rich compound, whose oxygen content reaches 49.98%. It doesn′t need extra oxygen in combustion process according to equation (1). In order to obtain the standard molar enthalpy of combustion for DNF, another idealized reaction equation (2) was adopted.
| $ {\rm{M(l) = }}\mathit{a}\;{{\rm{O}}_{\rm{2}}}({\rm{g}}) + \mathit{b}\;{\rm{C}}{{\rm{O}}_2}({\rm{g}}) + \mathit{c}\;{{\rm{N}}_2}({\rm{g}}) $ | (2) |
M=DNF: C2O5N4, a=2, b=1/2, c=2;
Meanwhile, in consideration of the rich nitro group in these compounds, idealized reaction equation (3) was employed. In the following reaction, NO2 was considered as the gas product instead of usual N2 for thermochemical calculation.
| $ {\rm{M(s)}} + \mathit{a}\;{{\rm{O}}_{\rm{2}}}({\rm{g}}) = \mathit{b}\;{\rm{C}}{{\rm{O}}_2}({\rm{g}}) + \mathit{c}\;{{\rm{H}}_2}{\rm{O(l)}} + \mathit{d}\;{{\rm{NO}}_2}({\rm{g}}) $ | (3) |
M=DAF:C2H4ON4, a=13/2, b=2, c=2, d=4;
DNF:C2O5N4, a=7/2, b=2, c=0, d=4;
DAAzF:C4H4O2N8, a=12, b=4, c=2, d=8;
The standard molar enthalpy of combustion (ΔcHmΘ) can be obtained in accordance with equations (4) and (5) from the constant-volume state to the constant-pressure state. The calculated results are listed in Table 2.
| $ {\Delta _{\rm{c}}}H_{\rm{m}}^\Theta = {\Delta _{\rm{c}}}{U_{\rm{m}}} + \Delta nRT $ | (4) |
| $ \Delta n = \sum {{n_i}({{\rm{products,}}\;{\rm{g}}})}-\sum {{n_i}({{\rm{reactants,}}\;{\rm{g}}})} $ | (5) |
| Tab.2 The thermodynamic values for the three furazan compounds at 298.15 K |
where ∑ni was the total molar amount of gases in products or reactants.
The standard molar enthalpy of formation(ΔfHmΘ) of the compound can be calculated by Hess′s law[19], according to the above thermochemical equations. Taking DAF for instance, the standard molar enthalpy of formation is:
| $ \begin{array}{l} {\Delta _{\rm{f}}}H_{\rm{m}}^\Theta ({\rm{DAF,}}\;{\rm{s}}) = 2{\Delta _{\rm{f}}}H_{\rm{m}}^\Theta ({\rm{C}}{{\rm{O}}_{\rm{2}}}{\rm{,}}\;{\rm{g}}) + 2{\Delta _{\rm{f}}}H_{\rm{m}}^\Theta ({{\rm{H}}_{\rm{2}}}{\rm{O,}}\;{\rm{l}}) - \\ {\Delta _{\rm{c}}}H_{\rm{m}}^\Theta ({\rm{DAF,}}\;{\rm{s}}) \end{array} $ | (6) |
where the standard molar enthalpies of formation for CO2(g) and H2O(l) recommended by CODATA, ΔfHmΘ(CO2, g)=(-393.51±0.13) kJ·mol-1, ΔfHmΘ(H2O, l)=(-285.83±0.042) kJ·mol-1 and ΔfHmΘ(NO2, g)=33.18 kJ·mol-1, were used to calculate the values of ΔfHmΘ[20-21]. The standard molar enthalpies of formation for the three compounds can be calculated and are listed in Table 2.
If N2 was considered as gas product, DAF has negative enthalpy of formation as (-57.1±11.9) kJ·mol-1, while DNF and DAAzF have positive enthalpy of formation as (295.3±6.0) kJ·mol-1 and (327.8±10.5) kJ·mol-1. Compared with amino group, nitro group has a greater effect on positive enthalpy of formation. Diazotization structure also contributes to positive enthalpy of formation. Moreover, if NO2 was considered as gas product, the value of enthalpy of formation of DAF is in close proximity to the literature value as 89.99 kJ·mol-1[22], which indicates that considering NO2 as gas product is feasible. Enthalpies of formation for DAF, DNF and DAAzF are (80.6±11.9), (437.9±6.0) kJ·mol-1 and (603.1±10.5) kJ·mol-1 respectively, and the change rule is consistent with the above.
3.3 Specific Heat CapacityThe continuous specific heat capacities of DAF, DNF and DAAzF were measured successively. Taking DAAzF as example, the measuring results are shown in Fig. 1. The fitted specific heat capacity equations for the three compounds are listed in Table 3, according to the automatic data processing software of Micro-DSC Ⅲ apparatus. Relevant specific heat capacities and standard molar heat capacities (cp, mΘ) of the three compounds at 298.15 K are also listed in Table 3. Specific heat capacities for DAF and DAAzF present linear relationship with temperature in the determining temperature range, but specific heat capacities of DNF presents obvious quadratic relationship with temperature. The specific heat capacities for DAF, DNF and DAAzF at 298.15 K are (1.4067±0.0012), (1.4797±0.0015), (1.1058±0.0010) J·g-1·K-1, respectively. The specific heat capacity of the three compounds decreases in the order of DNF>DAF>DAAzF. Nitro group makes a greater contribution to specific heat capacity than amino group, which is opposite to the change tendency of energy of combustion (J·g-1). The specific heat capacity of DAF is larger than that of DAAzF, indicating that diazotization structure may decrease the specific heat capacity of compound, but the values of DAF and DAAzF are still close to each other, which is consistent with the result of energy of combustion.
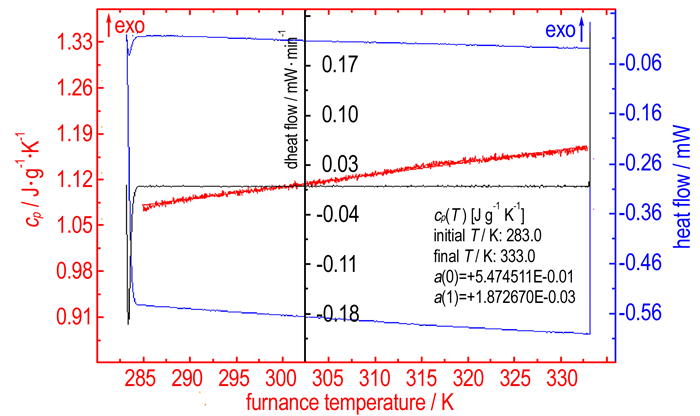
|
Fig.1 Determination result of the continuous specific heat capacity of DAAzF |
| Tab.3 Determination results of continuous specific heat capacity at 298.15 K |
(1) Energies of combustion of DAF, DNF and DAAzF were determined as (-13043±119), (-6863±37), (-12661±54) J·g-1, respectively. The energy of combustion tends to rise with the decrease of oxygen content in molecule. Amino group contributes to increase energy of combustion, but nitro group has the opposite effect. The corresponding standard molar enthalpies of formation (ΔfHmΘ) are (-57.1±11.9), (295.3±6.0), (327.8±10.5) kJ·mol-1 with N2 as gas product. When the gas product is NO2, the values of ΔfHmΘ are (80.6±11.9), (437.9±6.0), (603.1±10.5) kJ·mol-1, respectively.
(2) The standard molar heat capacities (cp, mΘ) of the three compounds are (140.8±0.1), (236.8±0.2), (216.9±0.2) J·mol-1·K-1, respectively. As for the specific heat capacity, the change rule is opposite to energy of combustion. In addition, diazotization has a faintly negative effect on energy of combustion and specific heat capacity.
| [1] |
LI Zhan-xiong, TANG Song-qing, OU Yu-xiang, et al. Synthesis status of furazano energetic Derivatives[J]. Chinese Journal of Energetic Materials(Hanneng Cailiao), 2002, 10(2): 59-65. |
| [2] |
Talawar M B, Sivabalan R, Senthilkumar N, et al. Synthesis, characterization and thermal studies on furazan-and tetrazine-based high energy materials[J]. Journal of Hazardous Materials, 2004, 113(1-3): 1125. |
| [3] |
GE Zhong-xue, WANG Xi-jie, JIANG Jun, et al. Synthesis of 3, 4-dinitrofurazan[J]. Chinese Journal of Synthetic Chemistry, 2008, 116(3): 260-263. |
| [4] |
Veauthier J M, Chavez D E, Tappan B C, et al. Synthesis and characterization of furazan energetics ADAAF and DOATF[J]. Journal of Energetic Materials, 2010, 28(3): 229-249. DOI:10.1080/07370651003601769 |
| [5] |
Chavez D E, Parrish D A, Leonard P. The synthesis and characterization of a new furazan heterocyclic System[J]. Synlett, 2012, 23(14): 2126-2128. DOI:10.1055/s-0032-1316704 |
| [6] |
Sheremetev A B, KulginaV O, Aleksandrova N S, et al. Dinitro trifurazans with oxy, azo, and azoxy bridges[J]. Propellants, Explosives, Pyrotechnics, 1998, 23(3): 142-149. DOI:10.1002/(ISSN)1521-4087 |
| [7] |
Chavez D, Hill S, Hiskey M. Preparation and explosive properties of azo-and azoxy-furazans[J]. Journal of Energetic Materials, 2000, 18(2-3): 219-236. DOI:10.1080/07370650008216121 |
| [8] |
GE Zhong-xue, LAI Wei-peng, LIAN Peng, et al. Predicting on the detonation performances of poly-furazans with oxy bridges[J]. Chinese Journal of Energetic Materials(Hanneng Cailiao), 2008, 16(3): 280-284. |
| [9] |
ZHANG Yu, WANG Bo-zhou, XU Kang-zhen, et al. Synthesis and characteristics of bis(nitrofurazano)furazan (BNFF), an insensitive material with high energy-density[J]. Propellants, Explosives, Pyrotechnics, 2014, 39(6): 809-814. DOI:10.1002/prep.201400057 |
| [10] |
ZHAI Lian-jie, WANG Bo-zhou, HUO Huan, et al. Synthesis, crystal structure and thermal behavior of 3, 4-Bis(3-nitrofurazan-4-oxy)furazan[J]. Chinese Journal of Energetic Materials(Hanneng Cailiao), 2015, 23(1): 18-22. |
| [11] |
Novikova T S, Melnikova T M, Kharitonova O V, et al. An effective method for the oxidation of aminofurazans to nitrofurazans[J]. Mendeleev Commun, 1994, 4(4): 138-140. DOI:10.1070/MC1994v004n04ABEH000386 |
| [12] |
LI Yu-bin, HUANG Hui, LI Jin-shan, et al. HMX based low sensitive high explosives containing DAAzF[J]. Chinese Journal of Energetic Materials(Hanneng Cailiao), 2008, 16(3): 244-246. |
| [13] |
Pagoria P F. A review of energy materialssynthesis[J]. Thermochimica Acta, 2002, 384(1): 187-204. |
| [14] |
Sikder A K, Sikder N. A review of advanced high performance, insensitive and thermally stable energetic materials emerging for military and space applications[J]. Journal of Hazardous Materials, 2004, 112(1-2): 1-15. DOI:10.1016/j.jhazmat.2004.04.003 |
| [15] |
SUN Qian, LI Yan-feng, XU Kang-zhen, et al. Crystal structure and enthalpy of combustion of AEFOX-7[J]. Chinese Journal of Energetic Materials(Hanneng Cailiao), 2015, 23(12): 1235-1239. |
| [16] |
Certificate of Analysis Standard Reference Material 39j Benzoic Acid Calorimetric Standard, NBS, Washington D C, 1995.
|
| [17] |
Marthada V K. The enthalpy of solution of SRM 1655 (KCl) in H2O[J]. Journal of Research of the National Bureau of Standards, 1980, 85(6): 467-471. DOI:10.6028/jres.085.027 |
| [18] |
Ditmars D A, Ishihara S, Chang S S. Enthalpy and heat-capacity standard reference material: synthetic sapphire ((δ)-Al2O3) from 10 to 2250 K[J]. Journal of Research of the National Bureau of Standards, 1982, 87(2): 159-163. DOI:10.6028/jres.087.012 |
| [19] |
Atkins P, Paula J D. Atkins′ Physical Chemistry (7th)[M]. Beijing: High Education Press, 2006.
|
| [20] |
Cox J D. CODATA recommended key values for thermodynamics, 1977 Report of the CODATA Task Group on key values for thermodynamics[J]. Journal of Chemical Thermodynamics, 1978, 10(10): 903-906. DOI:10.1016/0021-9614(78)90050-2 |
| [21] |
HU Rong-zu, ZHAO Feng-qi, GAO Hong-xu, et al. Fundamentals and application of calorimetry[M]. Beijing: Science Press, 2011.
|
| [22] |
TIAN De-yu, ZHAO Feng-qi, LIU Jian-hong. Handbook of Energetic Materials and the Related Compounds[M]. Beijing: National Defense Industry Press, 2011.
|
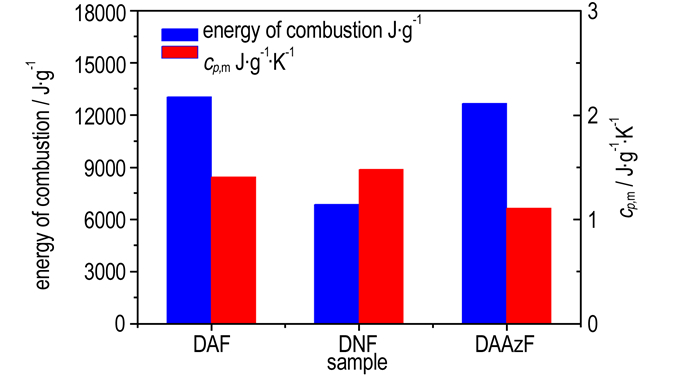
Energies of combustion for the three furazan compounds tend to rise with the decrease of oxygen content in molecule. Amino group contributes to increase energy of combustion, but nitro group has the opposite effect. To specific heat capacity, the change rule for the three compounds is opposite to energy of combustion.




